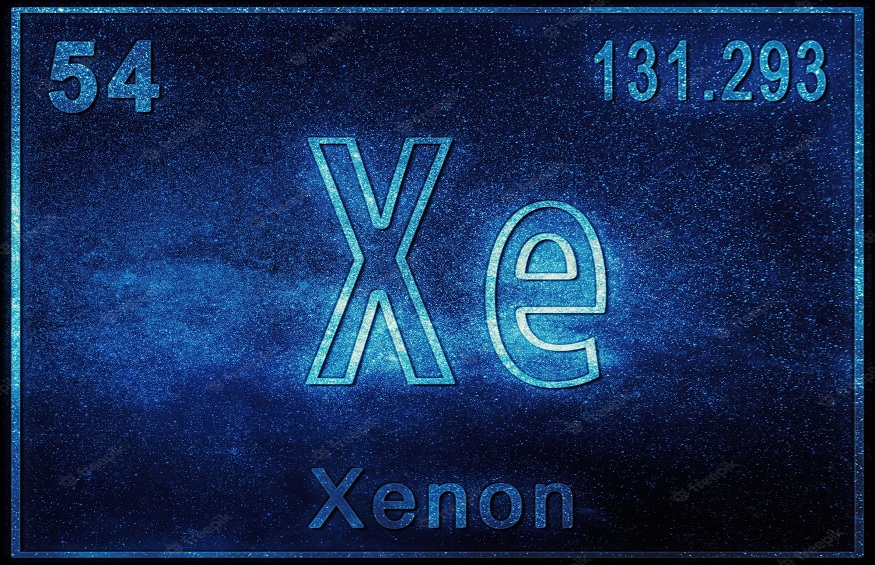
Chlorides of Potassium and Xenon
Most of the salts are solids. Both their melting and boiling points are high. Salts typically dissolve in water. Solutions of salts in water conduct electricity similarly to acids and bases. Salts are electrolytes, in other words. The ions in salt solutions cause them to conduct electricity. Compounds with ions are salts. A positively charged ion and a negatively charged ion make up every salt. For instance, sodium chloride (NaCl) is composed of negatively charged chloride ions and positively charged sodium ions.
A family of salts is considered to consist of salts with the same positive ions or the same negative ions. Because they both contain the same positively charged ions, sodium ions, Na+, sodium chloride (NaCl) and sodium sulphate (Na2SO4), both belong to the family of salts known as “sodium salts.” Similar to this, potassium chloride (KCI) and sodium chloride (NaCl) also come from the same salt family known as “chloride salts” because they both contain the same negatively charged ions, chloride ions. The number of salt families we can have equals the number of positively and negatively charged ions which make up the salts.
Applications of Xenon
The “starter gas” in high-pressure sodium lights is xenon. Of all the non-radioactive noble gases, it has the lowest thermal conductivity and ionisation potential. Being a noble gas, it has no effect on the chemical processes taking place inside the functioning lamp.
The first excimer laser produced stimulated emission at a wavelength of 176 nm by energising a xenon dimer (Xe2) with an electron beam. Additionally, xenon fluoride and xenon chloride have been utilised in excimer lasers. For example, the xenon chloride excimer laser has been used for specific dermatological procedures. A general anaesthetic utilising xenon has been used. Despite being costly, xenon anaesthetic devices are now available because improvements in xenon recovery and recycling have made them commercially viable.
Surgery can benefit from the profound anaesthetic properties of a combination of 20% oxygen and 80% xenon. Since xenon is entirely harmless and inflammable, it doesn’t carry any of the risks associated with more traditional anaesthetics like ether or ethylene. In bubble chambers, liquid xenon can be utilised to investigate the formation and characteristics of subatomic particles.
How Do Plants Use Potassium?
Plants take potassium ions and other nutrients from the soil through their roots. The plant absorbs many of these nutrients as ions. Potassium ions (K+) are used by plants to construct robust cell walls. They are able to grow tall and robust as a result. Additionally, it enables them to endure temperatures that are both hot and cold.
Different plants use different amounts of potassium ions. Apple trees require little water. Conversely, dandelions need a lot of potassium ions to survive. Applications for potassium and potassium compounds are numerous. Today, one of the most widely used industrial chemicals is potassium chloride. There are numerous other uses for potassium compounds, in addition to their long-standing use in fertiliser, soap and glass. They are used to create other compounds and to replace salt.



Average Rating